Capsule Export Packaging and Labelling Requirements: What Every Importer Needs to Know
When a consignment of empty hard gelatin capsules arrives at a port in Dubai, Lagos, or Nairobi, the first thing customs authorities and incoming QC teams check — before they open a single carton — is the outer labelling. Missing, incorrect, or non-compliant labelling is one of the most common causes of pharmaceutical import delays, quarantine holds, and even rejection of entire shipments.
This guide from Universe Capsules, an empty capsule manufacturer and exporter in India, explains exactly what should appear on capsule packaging and what documentation must accompany every export shipment.
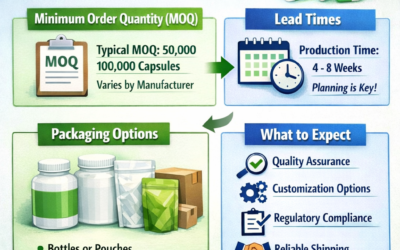
Mandatory carton labelling fields
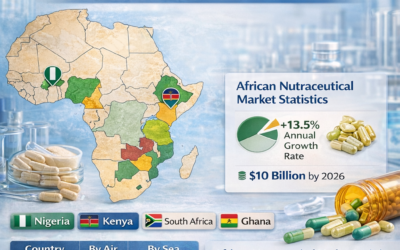
Requirements by destination market
- Full carton marking in English
- Halal certificate mandatory
- CoA with batch-specific results
- Certificate of origin
- SFDA import permit may be required — verify before shipment
- English labelling accepted
- NAFDAC registration may be required for industrial raw materials
- CoA and GMP certificate
- Phytosanitary / health certificate may be requested
- Pre-shipment inspection required in some cases
- English labelling accepted
- Halal cert strongly recommended
- CoA, GMP cert, TSE/BSE declaration
- Free trade zone imports (Dubai) have simpler requirements than direct pharma import
- English labelling accepted
- CoA and GMP certificate required
- Certificate of origin
- Relatively streamlined import process for pharmaceutical raw materials
What goes inside the carton — inner bag labelling
Each inner polythene bag must also be individually labelled — not just the outer carton. Inner bag labels should include at minimum: product name, size, colour, batch number, quantity per bag, and expiry date. In the event a carton is opened and bags are separated from the carton, the inner bag label must provide sufficient information to identify the product and batch without reference to the outer carton.
Common labelling mistakes that delay imports
- Batch number on carton does not match the CoA — the most common customs query
- Expiry date missing or ambiguous (e.g. "03/28" — is this March 2028 or the 3rd of the 28th?)
- Storage conditions absent from label — triggers additional inspection at some borders
- Country of origin missing — required for duty classification and certificate of origin compliance
- Halal mark present on label but Halal certificate not included in shipment documentation
- Quantity on label does not match packing list — discrepancy causes customs hold
Expecting full compliance from your supplier?
Universe Capsules exports with complete, compliant labelling and documentation — carton marking, batch CoA, Halal certificate, TSE/BSE declaration, certificate of origin, and commercial invoice — as standard on every shipment. No surprises at customs.
Frequently Asked Questions
What information must be on the outer carton of exported empty capsules?
Product name, capsule size and colour, batch number, manufacture date, expiry date, quantity, net weight, storage conditions, manufacturer name and address, and country of origin. Halal marking is additionally required for Middle East and African markets.
Do empty capsule cartons need to display storage conditions?
Yes. "Store at 15°C–25°C, 35%–65% RH, away from direct sunlight" must appear on both the outer carton and inner bag labels. This is a GMP requirement and essential for correct handling through the supply chain.
Is a Halal logo required on capsule packaging for Middle East markets?
For Halal-certified capsules destined for GCC and North African markets, the Halal certification mark from the recognised certifying body should appear on the packaging, with the certificate number referenced.
What languages are required on capsule export packaging?
English labelling is standard and accepted for pharmaceutical raw material imports in most global markets. Some countries may have retail-level language requirements, but for industrial capsule imports, English labelling with supporting certificates is generally sufficient.