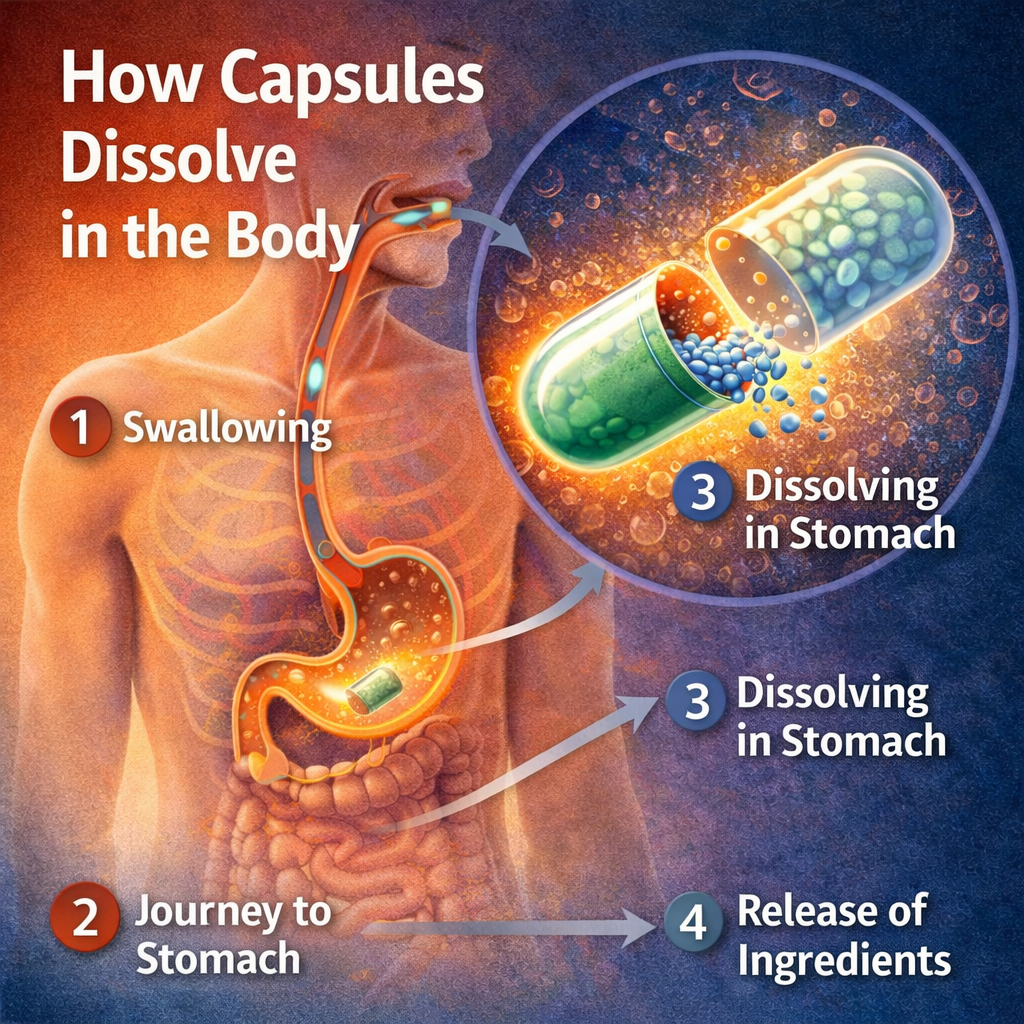
What Happens When a Gelatin Capsule Dissolves? Disintegration, Dissolution and Drug Release Explained
Every pharmaceutical professional knows that a capsule delivers a drug. But what actually happens in the 15 minutes after a patient swallows one? The sequence of disintegration, dissolution, and absorption is more complex — and more dependent on capsule shell quality — than most people appreciate.
This article from Universe Capsules, a hard gelatin capsule manufacturer in India, walks through the complete journey from swallow to absorption — and explains what it means for capsule selection and formulation development.
Stage 1 — Swallowing and gastric entry (0–2 minutes)
When a patient swallows a hard gelatin capsule, it travels down the oesophagus in 8–10 seconds and enters the stomach. Immediately, gastric fluid — a mixture of hydrochloric acid, enzymes, and water at approximately 37°C — begins to surround the capsule shell.
The capsule shell does not dissolve instantly. Gelatin is hydrophilic but requires time to absorb fluid and begin softening. In the first two minutes, the outer surface of the shell swells slightly as water penetrates the gelatin matrix. The capsule remains sealed during this period — no contents are released yet.
This is why capsule shell quality matters: a capsule with correct moisture content (13%–16%) has a gelatin matrix in the right physical state to absorb fluid uniformly. An over-dried capsule (below 13% moisture) has a denser, more cross-linked matrix that resists fluid penetration and slows disintegration.
Stage 2 — Shell softening and separation (2–5 minutes)
As the gelatin continues to absorb gastric fluid, the shell transitions from a glassy solid state to a rubbery, gel-like state. The cap and body begin to separate — not cleanly, but through a progressive softening of the locking groove that holds them together.
This is also the stage where capsule cross-linking can become a problem. When gelatin is exposed to certain reactive aldehydes — from formaldehyde-containing excipients, degrading lactose, or reactive API impurities — cross-links form in the gelatin matrix that make it resistant to swelling and dissolution. Heavily cross-linked gelatin capsules can take 45 minutes or longer to open, dramatically reducing drug bioavailability. This phenomenon, known as pellicle formation, is one of the more challenging stability problems in capsule formulation.
Stage 3 — Contents release and API dissolution (5–15 minutes)
Once the cap and body have separated sufficiently, the powder, granule, or pellet fill is exposed directly to gastric fluid. The rate at which the API then dissolves depends entirely on the formulation — not the capsule shell.
Key factors governing API dissolution from a capsule fill:
- Particle size of the API (smaller particles dissolve faster — higher surface area)
- API solubility in gastric and intestinal fluid
- Presence and type of disintegrant in the fill formulation
- Presence of surfactants to improve wetting of hydrophobic APIs
- Whether the fill contains modified-release pellets that are designed to resist gastric dissolution
Stage 4 — Absorption through the gastric and intestinal wall
Once dissolved, the API is absorbed through the gastric mucosa (for lipophilic, acid-stable drugs) or passes into the small intestine, where the vast majority of drug absorption occurs. The small intestine's large surface area — amplified by villi and microvilli — provides highly efficient absorption for most pharmaceutical APIs.
For capsule formulators, the key insight is this: the capsule shell's job ends at Stage 3. Everything that happens after the shell opens is determined by the fill formulation and the API's physicochemical properties. A high-quality capsule shell — one that disintegrates quickly and uniformly — creates the best possible conditions for Stage 3 to proceed efficiently.
What affects disintegration time — and what you can control
| Factor | Effect on disintegration | Controllable? |
|---|---|---|
| Capsule moisture content | Low moisture = slower, brittle shell; 13–16% optimal | Yes — specify on CoA |
| Gelatin cross-linking | Cross-linked shell resists fluid — much slower dissolution | Yes — avoid reactive excipients |
| Shell thickness | Thicker shell (larger size) = slightly slower | Partially — select correct size |
| Gastric pH | Higher pH (fed state, antacid use) slows gelatin dissolution | No — patient variable |
| Gastric motility | Faster motility = more agitation, faster disintegration | No — patient variable |
| Food in stomach | Fed state dilutes acid, slows shell softening slightly | Via dosing instructions |
| Capsule colour / pigment loading | High opacifier content can slightly reduce disintegration rate | Yes — balance with formulator |
Pharmacopoeia disintegration standard
The Indian Pharmacopoeia (IP), British Pharmacopoeia (BP), and United States Pharmacopeia (USP) all specify that hard gelatin capsules must disintegrate within 15 minutes when tested in simulated gastric fluid (0.1N HCl) at 37°C using a standard disintegration apparatus.
Universe Capsules tests every batch of empty hard gelatin capsules for disintegration time and includes the result on each batch Certificate of Analysis. All batches are required to disintegrate well within the 15-minute pharmacopoeia limit.
Need capsules with documented disintegration performance?
Universe Capsules provides a batch Certificate of Analysis with every shipment — including disintegration test results per IP/BP. Our hard gelatin capsules are manufactured to consistently disintegrate within pharmacopoeia limits.
Frequently Asked Questions
How long does a hard gelatin capsule take to dissolve?
Typically 3–15 minutes in gastric fluid at 37°C. The pharmacopoeia standard requires disintegration within 15 minutes. The exact time depends on capsule moisture content, shell thickness, and gastric conditions.
What is the difference between disintegration and dissolution?
Disintegration is the physical breaking apart of the capsule shell. Dissolution is the subsequent process of API particles dissolving into gastric fluid for absorption. Both must occur efficiently for good bioavailability.
Does food in the stomach affect capsule dissolution?
Yes, slightly. The fed state dilutes gastric acid and slows gastric emptying, which can marginally slow disintegration. For most drugs this is clinically insignificant, but it should be evaluated for APIs with narrow therapeutic windows.
Can a gelatin capsule be opened and contents taken without the shell?
Only if the formulation permits it. Capsules containing modified-release pellets or enteric-coated beads must not be opened as this destroys the release mechanism. Always check the product SmPC or consult a pharmacist first.