How Empty Hard Gelatin Capsules Are Manufactured: A Step-by-Step Guide
Have you ever wondered what goes into producing the tiny capsule shells that deliver pharmaceutical doses safely and precisely? The manufacturing of empty hard gelatin capsules is a highly controlled, technology-driven process — and getting it right is critical for the global pharmaceutical and nutraceutical supply chain. As a dedicated empty capsules manufacturer in India, Universe Capsules follows stringent IP and BP standards to produce pharma-grade capsule shells that meet international quality benchmarks.
This guide walks you through the complete capsule manufacturing process — from raw material selection to the finished product ready for your encapsulation line.
Why Manufacturing Quality Matters in Empty Capsule Production
Empty capsule shells are not passive containers. They directly influence:
- Drug dissolution rate and bioavailability
- Moisture protection of the API and excipients inside
- Compatibility with high-speed capsule filling machines
- Patient safety through TSE/BSE-free raw material sourcing
- Regulatory compliance for export across global markets
This is why every reputable capsule manufacturing company in India invests in state-of-the-art machinery, clean room infrastructure, and rigorous in-process quality controls at every stage.
Raw Materials Used in Hard Gelatin Capsule Manufacturing
The primary raw material for hard gelatin capsules is pharmaceutical-grade gelatin, derived from bovine or porcine collagen. It must be:
- TSE/BSE free — confirmed through supplier certification
- Compliant with IP, BP, USP, and EP pharmacopoeial standards
- Free from heavy metals, microbial contamination, and unacceptable color additives
Additional raw materials include purified water, permitted food-grade colorants (FD&C and D&C dyes), opacifying agents such as titanium dioxide, and preservatives where required. All incoming materials are subjected to rigorous incoming quality control (IQC) before entering production.
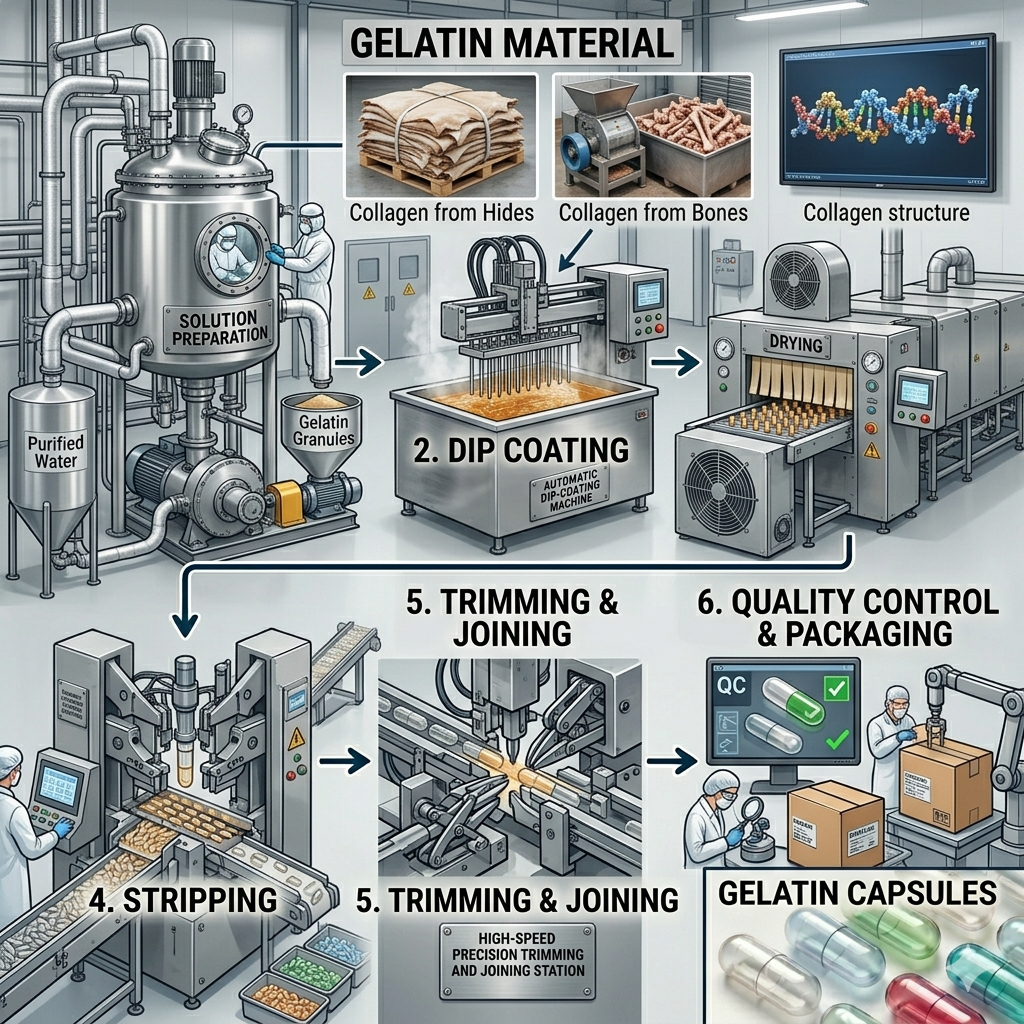
Step-by-Step Manufacturing Process of Empty Hard Gelatin Capsules
Step 1: Gelatin Dissolution and Dipping Solution Preparation
Pharmaceutical-grade gelatin is dissolved in purified water at controlled temperatures to create a uniform dipping solution. The concentration, viscosity, and temperature of this solution are precisely calibrated — as they directly determine the wall thickness and mechanical strength of the final capsule shell. Colorants and opacifiers are added at this stage under strict formulation controls.
Step 2: Pin Bar Dipping
Stainless steel pins — shaped to match the exact internal dimensions of the capsule body and cap — are dipped into the gelatin solution. This is one of the most technically sensitive steps in the process. The dipping speed, solution temperature, and pin surface condition must all be precisely controlled to ensure uniform film coating across every pin.
Universe Capsules uses modern pin-dipping machines capable of producing thousands of capsule halves per hour while maintaining dimensional consistency to within tight tolerances.
Step 3: Drying
After dipping, the gelatin-coated pins are passed through a series of carefully controlled drying kilns. Temperature, humidity, and airflow in each kiln section are independently monitored and adjusted. Proper drying is critical — under-drying leads to soft, fragile shells while over-drying causes brittleness and cracking. The target moisture content for finished hard gelatin capsules is 13–16%, as specified by international pharmacopoeias.
Step 4: Stripping
Once fully dried, the gelatin films are mechanically stripped from the steel pins. This is done using automated stripping mechanisms that apply uniform force to remove the capsule halves without distortion or damage. The stripped capsule bodies and caps are then conveyed to the next processing station.
Step 5: Trimming
Each capsule half — body and cap — is trimmed to the precise length specified for that capsule size. Trimming is performed by high-precision cutting blades that ensure dimensional uniformity. This step directly impacts how well the capsule closes on filling machines and how consistent fill weights will be during pharmaceutical manufacturing.
Step 6: Joining (Coupling)
The trimmed capsule bodies and caps are joined together in a pre-closed configuration for packaging and supply. This makes them easy to handle on automated capsule filling machines, which separate the two halves, fill them, and re-join them automatically during production.
Step 7: In-Process and Final Quality Control
Throughout the entire manufacturing process, samples are drawn at regular intervals for in-process quality checks. Final quality control testing includes:
- Dimensional inspection — length, diameter, wall thickness checked against specification
- Moisture content analysis — Karl Fischer titration or loss-on-drying method
- Disintegration testing — per pharmacopoeial requirements
- Microbial testing — total viable count, absence of specified pathogens
- Visual inspection — color uniformity, absence of defects (dents, holes, splits)
- TSE/BSE compliance documentation — batch-level certification provided
Only batches that pass all quality parameters are released for packaging and dispatch.
Step 8: Packaging and Dispatch
Approved capsules are counted, packed into polybags or cartons, and sealed under controlled humidity conditions to preserve moisture content during storage and transit. Each batch is accompanied by a Certificate of Analysis (CoA) covering all tested parameters. Export packaging complies with international pharmaceutical shipping standards.
Universe Capsules: Manufacturing Excellence in India
Universe Capsules Biotech Pvt. Ltd. is a dedicated empty hard gelatin capsule manufacturer in India producing capsule shells in sizes #000, 00, 0, 1, 2, 3, 4, and 5. Our manufacturing facility operates under WHO-GMP aligned practices with full traceability from raw material to finished product.
- ✔ Pharmaceutical-grade gelatin — TSE/BSE free, IP/BP compliant
- ✔ Available in plain, metallic, pearl, printed, and custom color combinations
- ✔ Compatible with all major high-speed capsule filling machines
- ✔ Batch-level CoA and regulatory documentation provided
- ✔ Supplying pharma companies across India and global markets
Learn more about our manufacturing capabilities or get in touch to discuss your capsule supply requirements.
Frequently Asked Questions (FAQ)
Q1: What raw materials are used to manufacture hard gelatin capsules?
The primary material is pharmaceutical-grade gelatin (bovine or porcine origin), combined with purified water, food-grade colorants, and opacifiers like titanium dioxide. All inputs must be TSE/BSE free and pharmacopoeially compliant.
Q2: What does TSE/BSE free mean?
TSE/BSE free certification confirms that the gelatin used is free from Transmissible Spongiform Encephalopathy and Bovine Spongiform Encephalopathy. This is a mandatory safety and regulatory requirement for pharmaceutical-grade capsule shells.
Q3: How many sizes does Universe Capsules manufacture?
Universe Capsules produces empty hard gelatin capsules in all standard sizes — #000, 00, 0, 1, 2, 3, 4, and 5 — for pharmaceutical and nutraceutical applications.
Q4: Are your capsules compatible with high-speed filling machines?
Yes. All capsules manufactured by Universe Capsules meet the dimensional standards required for compatibility with major high-speed encapsulation equipment used in the pharmaceutical industry.
Q5: Is a Certificate of Analysis provided with each batch?
Yes. Every dispatched batch comes with a full Certificate of Analysis (CoA) covering dimensional parameters, moisture content, microbial limits, and TSE/BSE compliance documentation.
Conclusion
The manufacturing of empty hard gelatin capsules is a precise, multi-step process where every stage — from raw material selection to final quality testing — determines the safety, performance, and reliability of the finished capsule shell. Universe Capsules brings together advanced manufacturing infrastructure, strict quality controls, and deep expertise to deliver pharma-grade capsule shells that meet the demands of modern pharmaceutical production in India and globally.
Explore our full product range or contact us for a quote.