How to Import Empty Hard Gelatin Capsules from India: A Step-by-Step Guide for Overseas Buyers
India is the world's second-largest pharmaceutical exporter, and its capsule manufacturing sector supplies buyers across the Middle East, Africa, Southeast Asia, and beyond. For overseas pharmaceutical manufacturers, nutraceutical companies, and Ayurvedic product exporters, sourcing empty hard gelatin capsules from India offers a compelling combination of quality, price, and supply reliability.
However, importing pharmaceutical raw materials across borders involves documentation requirements, regulatory clearances, and logistics decisions that first-time importers often find complex. This guide from Universe Capsules — an experienced capsule exporter from India — walks you through the process step by step.
Step 1: Qualify Your Supplier Before Anything Else
Before you think about shipping terms or import duties, the most important step is selecting a supplier who can consistently meet your quality and documentation requirements. An Indian capsule manufacturer exporting to regulated markets must be able to provide:
- Valid WHO-GMP certificate or Schedule M compliance
- Batch-specific Certificate of Analysis (CoA)
- TSE/BSE-free declaration with gelatin source traceability
- Halal certificate from a recognised body (for Middle East and African markets)
- Samples for filling trials on your equipment before bulk ordering
Rushing to the logistics stage before completing supplier qualification is the most common and costly mistake overseas buyers make. A capsule that fails your incoming QC after clearing customs costs far more than any freight saving.
Step 2: Understand the Regulatory Requirements in Your Country
Import regulations for empty hard gelatin capsules vary significantly depending on your country and how the capsules are classified — as pharmaceutical raw materials, pharmaceutical packaging, or finished pharmaceutical products.
Middle East (GCC countries)
In Saudi Arabia, the Saudi Food and Drug Authority (SFDA) regulates the import of pharmaceutical raw materials including empty capsules. Importers should verify current SFDA requirements before their first shipment, as registration or pre-approval may be required depending on end use. In the UAE, the Ministry of Health and Prevention (MOHAP) governs pharmaceutical imports, and requirements depend on whether capsules are used in local manufacturing or are being re-exported.
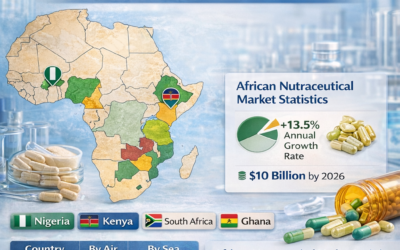
Nigeria
The National Agency for Food and Drug Administration and Control (NAFDAC) regulates pharmaceutical imports into Nigeria. Empty capsules intended for pharmaceutical manufacturing may require NAFDAC registration. Importers are advised to work with a NAFDAC-registered local agent and confirm registration requirements before placing their first bulk order.
Kenya and East Africa
The Pharmacy and Poisons Board (PPB) in Kenya regulates pharmaceutical raw material imports. Similar regulatory bodies govern imports in Tanzania (TFDA) and Uganda (NDA). Requirements are generally less complex than GCC or West African markets, but documentation standards — particularly GMP certification and CoA — must still be met.
Other markets
For any new import destination, consult a local regulatory affairs consultant or customs broker before committing to a supply arrangement. Regulatory requirements change, and penalties for non-compliant pharmaceutical imports can be severe.
Step 3: Confirm the HS Code and Import Duty
Empty hard gelatin capsules are typically classified under HS code 3913.90 (pharmaceutical products — other) in most international tariff systems, though some countries classify them under 3004.90 depending on how they treat pharmaceutical packaging versus pharmaceutical products. The classification affects the applicable import duty rate.
Work with a licensed customs broker in your country to confirm the correct HS code before your first shipment. Mis-classification is a common cause of import delays and unexpected duty charges. Some markets offer zero or reduced duties on pharmaceutical manufacturing inputs — your broker can advise on applicable exemptions.
Step 4: Agree on Shipping Terms (Incoterms)
When placing your order with an Indian capsule manufacturer, you need to agree on shipping terms — known as Incoterms — which define who is responsible for freight, insurance, and customs clearance at each stage of the journey.
| Incoterm | What it means for you | Best for |
|---|---|---|
| EXW (Ex Works) | You arrange all freight from the supplier's factory gate | Buyers with established freight forwarders in India |
| FOB (Free on Board) | Supplier loads goods at Indian port; you arrange onward shipping | Most common for first-time importers |
| CIF (Cost, Insurance, Freight) | Supplier arranges shipping and insurance to your destination port | Buyers who want simplicity; less control over freight cost |
| DDP (Delivered Duty Paid) | Supplier handles everything including import duties at your end | First-time importers wanting full simplicity; usually higher price |
For most first-time importers from the Middle East or Africa, FOB or CIF terms with a reputable Indian freight forwarder are the most practical starting point.
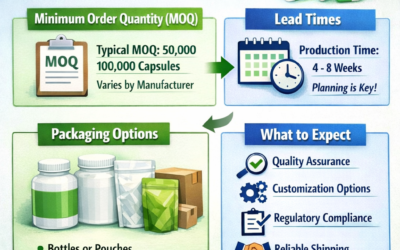
Step 5: Confirm the Documentation Package
A complete export documentation package from a professional Indian capsule manufacturer should include all of the following:
| Document | Purpose |
|---|---|
| Commercial invoice | Customs valuation and duty calculation |
| Packing list | Details of cartons, weights, and capsule quantities per size |
| Bill of lading / airway bill | Proof of shipment and title transfer |
| Certificate of origin | Confirms goods manufactured in India; may attract preferential duties |
| Certificate of analysis (CoA) | Batch quality confirmation; required by most pharma regulators |
| GMP certificate | Confirms manufacturer's regulatory compliance |
| TSE/BSE-free declaration | Required by most regulated pharmaceutical markets |
| Halal certificate | Required for GCC, North Africa, and Muslim-majority markets |
| Health certificate | May be required by specific destination countries |
Request a sample documentation package from your supplier before placing your first order. A supplier who cannot produce complete, accurate documentation will create problems at customs clearance that delay your production schedule.
Step 6: Plan Your Storage Before the Shipment Arrives
Empty hard gelatin capsules require controlled storage conditions — 15°C to 25°C, 35% to 65% relative humidity. Before your first shipment arrives, confirm that your receiving warehouse or storage facility can maintain these conditions. In tropical climates common across Africa and parts of the Middle East, uncontrolled warehouses will quickly degrade capsule quality and render the shipment unusable.
Plan to move incoming capsules to climate-controlled storage within 24 hours of receipt. Conduct incoming quality control inspection — visual check, dimensional check, and filling trial — on a sample from each batch before committing the full delivery to production.
Step 7: Build a Long-Term Supply Relationship
Pharmaceutical manufacturing depends on supply continuity. Once you have qualified a supplier and successfully received your first shipment, invest in building a long-term supply relationship rather than re-tendering every order. Benefits of a stable supplier relationship include priority production scheduling, consistent batch-to-batch quality, and faster resolution of any quality queries.
Universe Capsules works with buyers across the Middle East and Africa on a repeat supply basis, providing advance production scheduling, consistent documentation, and direct technical support for filling machine compatibility queries.
Ready to start importing from Universe Capsules?
We export pharma-grade empty hard gelatin capsules to buyers in the Middle East, Africa, and Asia. Our standard export package includes WHO-GMP certificate, batch CoA, TSE/BSE-free declaration, Halal certificate, and full shipping documentation. Samples available before bulk order.
Frequently Asked Questions
What documents are required to import empty capsules from India?
Standard documentation includes a commercial invoice, packing list, certificate of origin, bill of lading, certificate of analysis, and GMP certificate. For Middle East markets, a Halal certificate and country-specific health certificate are also typically required. Markets such as Nigeria and Saudi Arabia may require product registration before import.
What is the HS code for importing empty gelatin capsules from India?
Empty hard gelatin capsules are typically classified under HS code 3913.90. Some countries may classify them under 3004.90. Always confirm the correct HS code with your customs broker before shipment to avoid clearance delays or unexpected duty charges.
Is prior registration required to import capsules into Middle East countries?
Requirements vary by country. Saudi Arabia (SFDA) and UAE (MOHAP) have specific requirements depending on end use. Always consult the relevant national regulatory authority or a local regulatory affairs consultant before your first import.
How are empty capsules shipped from India — by sea or air?
Most bulk shipments travel by sea freight. Sea transit from India to Middle East ports takes 7–15 days. Air freight is available for urgent orders or samples but is significantly more expensive. Most regular buyers use sea freight as their primary shipping mode.
Can empty gelatin capsules be imported duty-free into African countries?
Import duties vary by country and HS classification. Some African countries offer reduced duties on pharmaceutical manufacturing inputs under specific trade agreements. Consult a local customs broker to determine applicable duty rates and exemption eligibility for your specific market.