How to Store Empty Hard Gelatin Capsules: A Complete Guide for Pharma Manufacturers
Improper storage of empty hard gelatin capsules is one of the most common and costly mistakes in pharmaceutical manufacturing. Capsules that arrive in perfect condition can become brittle, sticky, or deformed before they ever reach a filling machine — simply due to incorrect warehouse conditions.
This guide from Universe Capsules, a leading empty hard gelatin capsule manufacturer in India, covers everything pharmaceutical manufacturers, contract packagers, and bulk importers need to know about correct capsule storage.
Why Storage Conditions Matter for Gelatin Capsules
Hard gelatin capsules are made from pharmaceutical-grade gelatin — a protein-based material that is highly sensitive to both moisture and temperature. Unlike plastic or metal containers, gelatin shells actively exchange moisture with their surrounding environment. This means the storage atmosphere directly determines the physical state of every capsule in your inventory.
Poor storage leads to:
- Capsules sticking together, causing jams on automatic filling machines
- Brittle caps that crack or split when handled, increasing waste and rejection rates
- Dimensional changes affecting the fit between cap and body, leading to poor seals
- Microbial growth in high-humidity conditions, risking contamination
- Failed incoming quality checks and delayed production schedules
For pharmaceutical buyers in India, the Middle East, and Africa — where ambient temperatures and humidity levels are frequently extreme — correct storage is not optional. It is a GMP requirement.
The Two Critical Parameters: Temperature and Humidity
Temperature
Empty hard gelatin capsules must be stored at temperatures between 15°C and 25°C. This is the range at which gelatin maintains its correct mechanical properties — firm enough to run on filling equipment, yet flexible enough to resist cracking under normal handling.
| Temperature | Effect on capsules | Risk |
|---|---|---|
| Below 10°C | Gelatin becomes brittle | Cracking, splitting during filling |
| 15°C – 25°C | Optimal mechanical properties | None — ideal range |
| 26°C – 30°C | Gradual softening begins | Increased sticking, slower filling |
| Above 30°C | Significant softening and deformation | Capsules stick, seal poorly, reject |
Relative humidity (RH)
The recommended relative humidity range for storing empty hard gelatin capsules is 35% to 65% RH. Gelatin capsules contain a controlled amount of residual moisture — typically 13%–16% by weight — which keeps them in the correct equilibrium state. Humidity outside the safe range disrupts this equilibrium.
| Humidity (RH) | Effect on capsules | Risk |
|---|---|---|
| Below 35% | Capsules lose moisture, become brittle | Cracking, static build-up, poor filling |
| 35% – 65% | Stable moisture equilibrium | None — ideal range |
| Above 65% | Capsules absorb excess moisture, soften | Sticking, denting, microbial risk |
| Above 75% | Visible deformation, surface tackiness | Total batch rejection likely |
Storage Requirements by Region
Pharmaceutical manufacturers in different climate zones face different challenges when storing empty hard gelatin capsules.
India (tropical and semi-arid regions)
Most Indian pharmaceutical manufacturing hubs — including Indore, Hyderabad, Ahmedabad, and Mumbai — experience temperatures regularly exceeding 35°C in summer months, with monsoon humidity often above 80% RH. Air-conditioned, dehumidified storage rooms are mandatory for maintaining capsule integrity year-round.
Middle East (GCC countries)
Saudi Arabia, UAE, Kuwait, and surrounding markets present the opposite challenge: extreme heat combined with dry, low-humidity conditions in summer, and elevated humidity in coastal areas. Both extremes threaten capsule quality. Importers receiving bulk shipments should move capsules to climate-controlled storage within 24 hours of receipt.
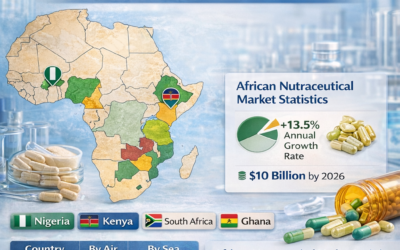
Africa (West and East Africa)
Markets such as Nigeria, Kenya, Ghana, and Tanzania experience high humidity throughout much of the year. Warehouses without adequate air conditioning and dehumidification routinely cause capsule degradation. Importers should specify ICH Zone IVb storage conditions (30°C / 75% RH stability testing) when sourcing capsules for these markets — and ensure their own facilities stay well within the 35%–65% RH storage range.
Practical Storage Guidelines
1. Store in the original sealed packaging
Universe Capsules supplies empty hard gelatin capsules in moisture-barrier bags or polythene-lined cartons that are specifically designed to maintain the correct moisture equilibrium during transit and storage. Do not open packaging until the capsules are required for production. Opened bags should never be left exposed for more than a few hours.
2. Keep away from direct sunlight and heat sources
UV exposure and radiant heat from windows, machinery, or lighting accelerates moisture loss and can cause colour changes in capsule shells. Store cartons away from external walls, windows, and any heat-generating equipment.
3. Elevate stock off the floor
Store capsule cartons on pallets or shelving units, a minimum of 10 cm off the floor. Floor storage risks moisture ingress, pest contact, and temperature fluctuations from cold concrete surfaces.
4. Implement FIFO stock rotation
Always use the oldest stock first (First In, First Out). Label each incoming batch with the receipt date and manufacturing batch number. Running expired or near-expiry stock increases the risk of capsule quality failures during production.
5. Monitor and log conditions daily
Install calibrated temperature and humidity data loggers in your capsule storage area. Most GMP guidelines require documented evidence of storage conditions for each batch. Acceptable excursions should be defined in your SOPs, and any out-of-range reading should trigger a batch review.
6. Equilibrate before filling
If capsules have been stored in cooler or drier conditions, allow them to equilibrate to the filling room environment (typically 20°C–25°C, 40%–55% RH) for at least 24 hours before use. Capsules moved directly from cold storage to a warm filling room are more prone to dimensional changes and splitting.
Signs That Capsules Have Been Stored Incorrectly
Before committing any batch of empty capsules to production, conduct a visual and physical inspection. Discard or quarantine any capsules showing:
- Visible sticking or clumping between shells
- Surface tackiness or a dull, matte finish (normal capsules have a slight sheen)
- Cracking or splitting at the rim or body
- Colour change or dark spots suggesting moisture damage
- Any odour, which may indicate microbial activity
Sourcing empty hard gelatin capsules for your facility?
Universe Capsules manufactures and exports pharma-grade empty hard gelatin capsules with full documentation including certificate of analysis, storage guidelines, and batch traceability. We supply manufacturers across India, the Middle East, and Africa.
Frequently Asked Questions
What is the ideal temperature for storing empty hard gelatin capsules?
Empty hard gelatin capsules should be stored at temperatures between 15°C and 25°C. Temperatures above 30°C can cause capsules to soften and deform, while temperatures below 10°C can make them brittle and prone to cracking during filling.
What humidity level is safe for gelatin capsule storage?
The recommended relative humidity for storing empty hard gelatin capsules is 35%–65% RH. Humidity above 65% causes capsules to absorb moisture, become soft, and stick together. Below 35%, they become brittle and crack during filling.
Can empty gelatin capsules be stored in a warehouse without climate control?
It is not recommended, particularly in tropical or high-humidity regions such as West Africa, South Asia, and coastal Gulf markets. Climate-controlled storage is essential to maintain capsule integrity and shelf life.
How long can empty hard gelatin capsules be stored?
When stored correctly (15°C–25°C, 35%–65% RH), empty hard gelatin capsules have a shelf life of 24–36 months from manufacture. Always verify the specific batch expiry date on the certificate of analysis provided by your supplier.
Should empty capsules be kept in their original packaging until use?
Yes. Original packaging maintains the correct moisture equilibrium for the capsules inside. Opened bags should be used promptly. Partial bags left open in a filling room will degrade faster and increase rejection rates during production.