Capsule vs Tablet: Which Is the Right Dosage Form for Your Formulation?
Two dosage forms dominate solid oral pharmaceutical manufacturing: capsules and tablets. Both deliver APIs orally. Both are manufactured at industrial scale. But they have very different development timelines, manufacturing requirements, cost profiles, and patient characteristics — and choosing the wrong one at the start of a project can cost months of reformulation work later.
This guide from Universe Capsules, a hard gelatin capsule manufacturer in India, provides a direct, practical comparison to help pharmaceutical developers and buyers make the right choice for their formulation.
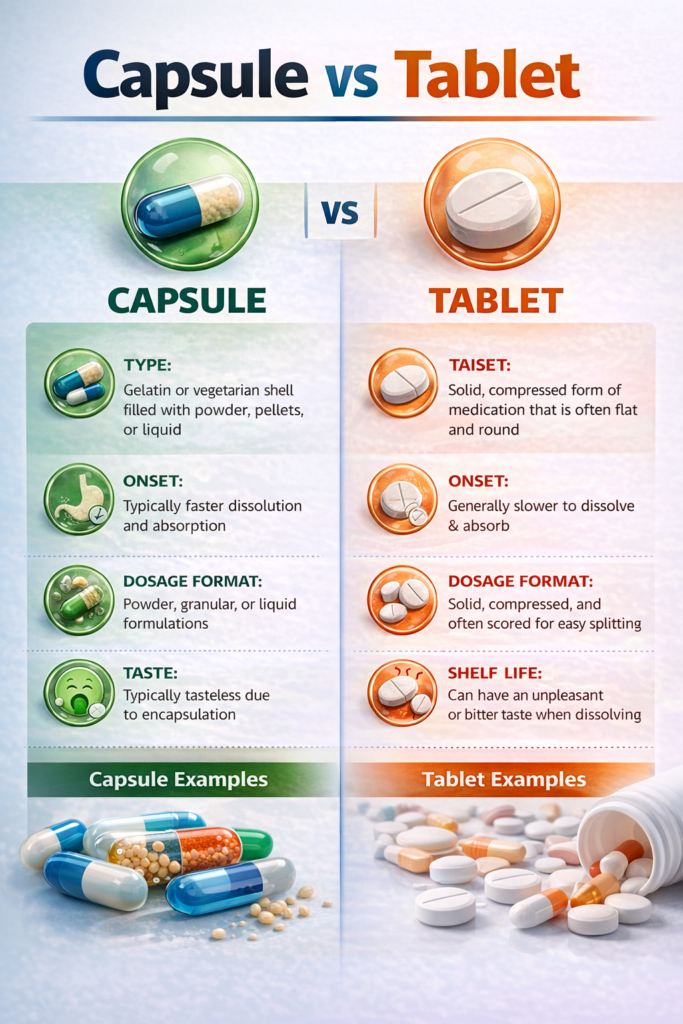
Hard gelatin capsule
Two-piece shell. Filled with powder, granules, or pellets. No compression required. Fast to develop.
Tablet
Compressed powder blend. Can be coated, scored, or shaped. Higher tooling complexity, lower per-unit cost at scale.
Head-to-head comparison
| Factor | Capsule | Tablet |
|---|---|---|
| Development time | ⚡ Weeks | Several months |
| Manufacturing equipment | Filling machine + capsule supply | Tablet press (common) |
| Small batch cost | Lower — no compression tooling | Higher tooling investment |
| High-volume unit cost | Moderate | Lower at very high volumes |
| Heat-sensitive APIs | Excellent — no heat or compression | Compression force may degrade API |
| Hygroscopic APIs | Caution — gelatin absorbs moisture | Better with film-coating protection |
| Patient swallowability | Generally preferred — smooth shell | Some patients find tablets harder |
| Dose splitting | Not possible | Scored tablets allow dose splitting |
| Taste masking | Excellent — shell isolates contents | Requires film or sugar coating |
| Modified release | Excellent with pellet/multiparticulate fills | Good with matrix or coated systems |
| Brand differentiation | Colours, printing, metallic/pearl finish | Shape, colour, embossing |
| Regulatory precedent | Extensive global precedent | Extensive global precedent |
When capsules are clearly the better choice
- Early-stage development where formulation speed is critical
- Heat or moisture-sensitive APIs that cannot tolerate compression
- Combination products with multiple APIs or pellet types
- Nutraceuticals, herbal powders, and Ayurvedic products
- Products where strong taste or odour masking is needed
- Premium or branded products where visual differentiation matters
- Clinical trial supplies requiring flexible, adjustable fill weights
When tablets may be the better choice
- Very high-volume commercial products where per-unit cost is paramount
- Products where patients need dose splitting capability
- APIs that are stable under compression with favourable flow properties
- Products already registered as tablets where switching would require regulatory re-filing
- Very low-dose APIs where weight uniformity of compressed tablets is superior
Ready to source capsules for your formulation?
Universe Capsules manufactures empty hard gelatin capsules in all standard sizes, available plain, printed, metallic, and pearl finish. We supply pharmaceutical manufacturers and nutraceutical companies across India, the Middle East, and Africa.
Frequently Asked Questions
Are capsules or tablets better for patient compliance?
Patients generally prefer capsules for ease of swallowing, particularly elderly patients. However, tablets can be scored for dose splitting, which capsules cannot, giving tablets an advantage where dose flexibility is needed.
Are capsules more expensive to manufacture than tablets?
For small to medium batches, capsules are generally cheaper due to simpler equipment and no compression development. At very large commercial scales, tablets can become more cost-effective due to higher press throughput.
Which is faster to develop — capsules or tablets?
Capsules are significantly faster. A hard gelatin capsule formulation can move from API to clinical trial supply in weeks. Tablet development typically requires months of compression, granulation, and coating studies.
Can any API be formulated as a capsule?
Most solid APIs are compatible. Exceptions include highly hygroscopic APIs, APIs incompatible with gelatin, liquid or oily APIs (which require softgels), and very low-dose APIs where filling weight variation may be unacceptable.
Which dosage form is better for modified release formulations?
Both support modified release. Capsules are preferred for multiparticulate systems using coated pellets; tablets are common for matrix-based extended release. The choice depends on release mechanism and manufacturing capabilities.