Encapsulating Probiotics: A Complete Guide to Probiotic Capsule Formulation
The global probiotic supplement market is one of the fastest-growing segments in nutraceuticals — and capsules are the preferred delivery format for the majority of probiotic products worldwide. But probiotic encapsulation is significantly more technically demanding than standard vitamin or mineral capsule formulation. Living bacterial cells are sensitive to heat, moisture, oxygen, and pH in ways that inactive pharmaceutical ingredients simply are not.
This guide from Universe Capsules, a hard gelatin capsule manufacturer in India, covers everything formulators and nutraceutical manufacturers need to know before starting a probiotic capsule project.
Why probiotic capsule formulation is uniquely challenging
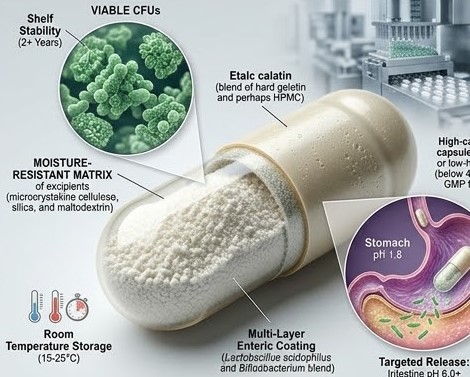
Probiotic microorganisms — primarily Lactobacillus and Bifidobacterium species — are living organisms that can die in unfavourable conditions. Unlike a chemical API whose stability is governed by chemical degradation pathways, a probiotic's viability depends on moisture, temperature, oxygen exposure, and pH. The formulator's job is to create conditions that keep the organisms alive from the moment of manufacture to the last capsule consumed — which may be 18–24 months later.
The critical challenge is moisture. Probiotic organisms are stabilised by lyophilisation (freeze-drying), which removes water and puts cells into a dormant state. Any reintroduction of moisture — from the capsule shell, from excipients, or from environmental exposure through inadequate packaging — begins to reactivate cells that then die from lack of nutrients. Managing moisture is therefore the central technical challenge in probiotic capsule formulation.
Capsule type selection for probiotics
Hard gelatin capsules — suitable with precautions
Hard gelatin capsules are widely used for probiotic products, particularly for strains with moderate moisture stability. The key precaution is managing the moisture transfer potential between the gelatin shell (13–16% moisture) and the probiotic powder fill. Formulation strategies include using moisture-absorbing excipients in the fill (microcrystalline cellulose, anhydrous lactose, silicon dioxide) that act as moisture sinks before moisture reaches the probiotic organisms.
HPMC capsules — preferred for sensitive strains
HPMC capsules with lower moisture content (4–6%) present a lower moisture transfer risk to hygroscopic probiotic fills. For highly moisture-sensitive strains, HPMC capsules are the preferred choice despite higher cost. The lower moisture content of the shell means less moisture available to migrate into the fill during storage.
CFU overage and end-of-shelf-life labelling
This is one of the most commercially important aspects of probiotic capsule formulation. Regulatory and consumer expectations require that the CFU count on the label be the count at end of shelf life — not at manufacture. Since probiotic organisms die off during storage at a rate depending on conditions, formulators must add a sufficient overage at manufacture to ensure label claims are still met at expiry.
Typical overage factors:
- Room-temperature stable formulation: 2–3× label claim at manufacture
- Refrigerated storage: 1.2–1.5× label claim at manufacture
- High-temperature or humid market: up to 5× overage may be required
Stability studies at the intended storage conditions are essential to determine the appropriate overage for your specific strain and formulation. Do not use literature die-off rates — measure your actual formulation.
Excipient selection for probiotic capsule fills
| Excipient | Function | Typical level | Probiotic suitability |
|---|---|---|---|
| Microcrystalline cellulose (MCC) | Diluent, moisture absorber | 20–40% | Excellent — low moisture, good flow |
| Anhydrous lactose | Diluent, flow aid | 20–40% | Good — but check strain lactose tolerance |
| Colloidal silicon dioxide | Glidant, moisture absorber | 0.5–2% | Excellent — essential for flow and moisture control |
| Magnesium stearate | Lubricant | 0.5–1% | Use sparingly — can reduce viability at high levels |
| Maltodextrin | Protective carrier (from lyophilisation) | 10–30% | Excellent — standard cryoprotectant |
| Inulin / FOS | Prebiotic synbiotic effect | 5–20% | Good — hygroscopic, monitor moisture |
Storage and secondary packaging requirements
Probiotic capsule products have more demanding packaging requirements than most nutraceuticals:
- Alu-alu blister: Highest moisture and oxygen barrier — preferred for sensitive strains and tropical markets
- HDPE bottle with desiccant and induction seal: Standard for room-temperature shelf-stable products; desiccant canister is non-negotiable
- Storage temperature: Most shelf-stable probiotics: 15°C–25°C, away from direct sunlight. Refrigerated products: 2°C–8°C throughout the supply chain
Need capsules for your probiotic formulation?
Universe Capsules manufactures hard gelatin and can advise on capsule selection for probiotic applications. We supply nutraceutical manufacturers across India, the Middle East, and Africa with full GMP documentation.
Frequently Asked Questions
Are hard gelatin capsules suitable for probiotics?
Yes, with moisture management precautions. Use moisture-absorbing excipients, fill in low-humidity conditions (below 40% RH), and use moisture-barrier secondary packaging. For highly sensitive strains, HPMC capsules with lower moisture content are preferred.
What CFU count is standard for probiotic capsules?
10–20 billion CFU is most common for general wellness products. Label claim must be end-of-shelf-life count — manufacturers add 2–5× overage at manufacture to account for die-off during storage.
Do probiotic capsules need refrigeration?
Depends on the strain and formulation. Properly lyophilised strains with moisture protection can achieve room-temperature stability for 12–24 months. Stability studies at intended storage conditions are essential to confirm viability through shelf life.
What capsule size is best for probiotic formulations?
Size 0 or 00 — probiotic powders are typically low density (0.3–0.5 g/ml), requiring larger capsule volumes. Size 00 is often preferred for high CFU-count products requiring larger total powder mass.