Pharmaceutical Capsule Supply Chain Risk Management: Lessons from Post-COVID Procurement
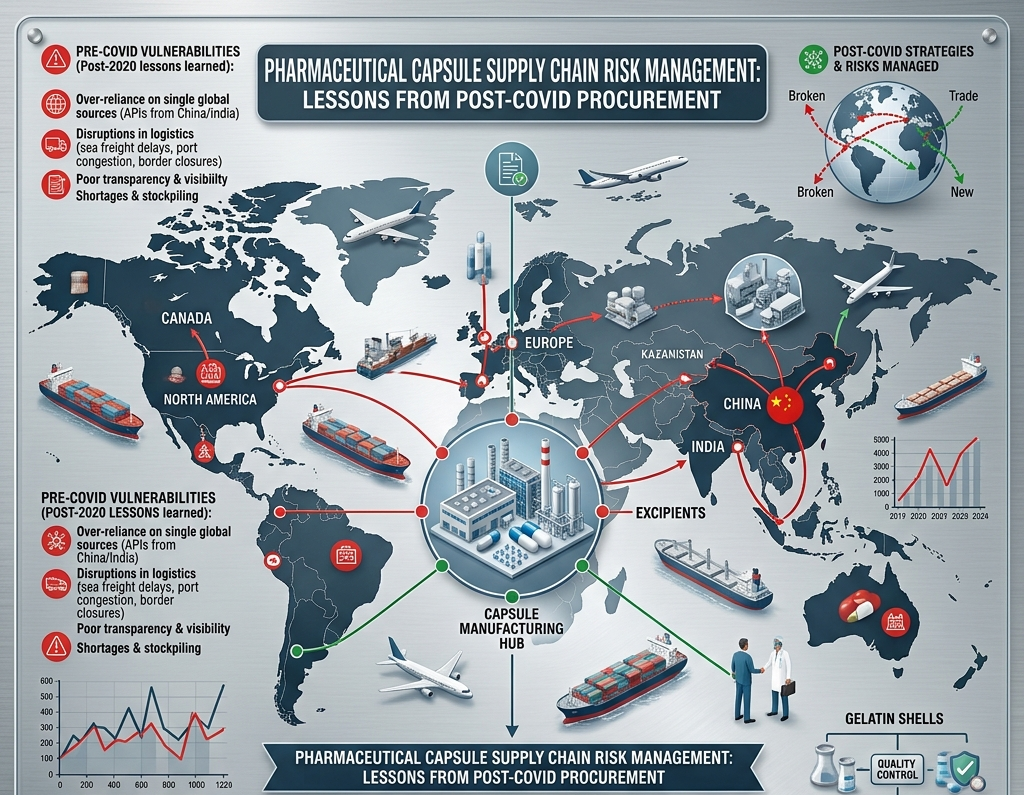
Before 2020, many pharmaceutical manufacturers ran lean, single-source raw material procurement without much thought of disruption. COVID-19 changed everything. Factory shutdowns, port closures, freight rate spikes, and demand surges exposed the fragility of just-in-time pharmaceutical supply chains globally — and empty capsule shells were among the first materials to run short.
This guide from Universe Capsules, a hard gelatin capsule manufacturer in India, shares a practical supply chain risk framework for pharmaceutical buyers who want to ensure production continuity.
The five principles of capsule supply chain resilience
1. Safety stock — your first line of defence
Maintaining adequate safety stock is the single most effective supply chain risk control. Industry best practice for pharmaceutical capsule shells is 8–12 weeks of average consumption in controlled storage. This buffer absorbs: supplier production delays, shipping transit variability, customs clearance delays, and the time needed to switch to an alternative supplier if required. Review your safety stock level quarterly against current lead time and consumption data.
2. Dual sourcing — eliminate single-supplier dependency
Single-source dependency is the highest-impact supply chain vulnerability. If your sole capsule supplier faces a production shutdown, quality recall, or regulatory suspension, your production line stops. Qualifying a second approved supplier for each capsule specification — even if the second source is used for only 10–20% of routine volume — provides a switchover option that can be activated rapidly in a disruption scenario.
3. Supplier financial and operational health monitoring
Suppliers face their own supply chain pressures. A gelatin supplier disruption can cascade to your capsule manufacturer within weeks. Periodically review your key suppliers' operational stability: visit facilities where possible, review quality audit outcomes, and ask suppliers about their own raw material security. A supplier who cannot answer confidently about their gelatin source security is a risk.
4. Long-term supply agreements
Spot purchasing exposes buyers to both price volatility and supply availability risk. Where volume justifies it, annual supply agreements with committed quarterly volumes give both parties planning certainty — your supplier can plan production capacity; you secure priority supply and typically more stable pricing. Forward agreements become especially valuable during industry-wide demand surges.
5. Geographic diversification of suppliers
Concentrating all capsule sourcing in one supplier, one industrial zone, or one country creates exposure to regional disruptions — floods, port strikes, regulatory events, or geopolitical developments. Maintaining at least one qualified supplier in a different geographic cluster within India reduces this exposure meaningfully.
Safety stock calculation guide
| Destination / route | Sea transit from India | Recommended safety stock | Rationale |
|---|---|---|---|
| Dubai / GCC | 5–9 days | 8 weeks | Fast route allows leaner buffer |
| East Africa (Mombasa) | 14–18 days | 10 weeks | Moderate transit; allow for customs delay |
| West Africa (Lagos) | 22–28 days | 14 weeks | Long transit + port congestion risk |
| India (domestic) | 2–5 days road | 6–8 weeks | Domestic risk mainly production delays |
| South Asia (Sri Lanka, Bangladesh) | 5–10 days | 8 weeks | Standard buffer for this transit time |
Build supply chain resilience with Universe Capsules
Universe Capsules offers annual supply agreements, confirmed lead times, advance production scheduling, and consistent batch-to-batch quality — all of which support your supply chain resilience strategy. We export to buyers across the Middle East and Africa.
Frequently Asked Questions
What are the main supply chain risks for capsule buyers?
Single-source dependency, shipping disruptions, quality failures requiring emergency re-sourcing, supplier capacity constraints during peak demand, and freight rate volatility. All were exposed simultaneously during COVID-19.
How much safety stock should manufacturers carry for capsule shells?
8–12 weeks at average consumption for most markets. Up to 14–16 weeks for West Africa given longer sea transit and port congestion risk. Rotate on FIFO basis in controlled storage conditions.
What is dual sourcing for pharmaceutical raw materials?
Maintaining two qualified, approved suppliers for the same raw material. If one faces a disruption, production continues from the second source. Requires qualifying both suppliers through full QC and regulatory approval processes.
How did COVID-19 affect pharmaceutical capsule supply chains?
Factory shutdowns, port closures, freight rate spikes, and demand surges hit simultaneously. Manufacturers with diverse suppliers, strategic safety stocks, and strong supplier relationships maintained continuity; single-source just-in-time buyers faced severe shortages.